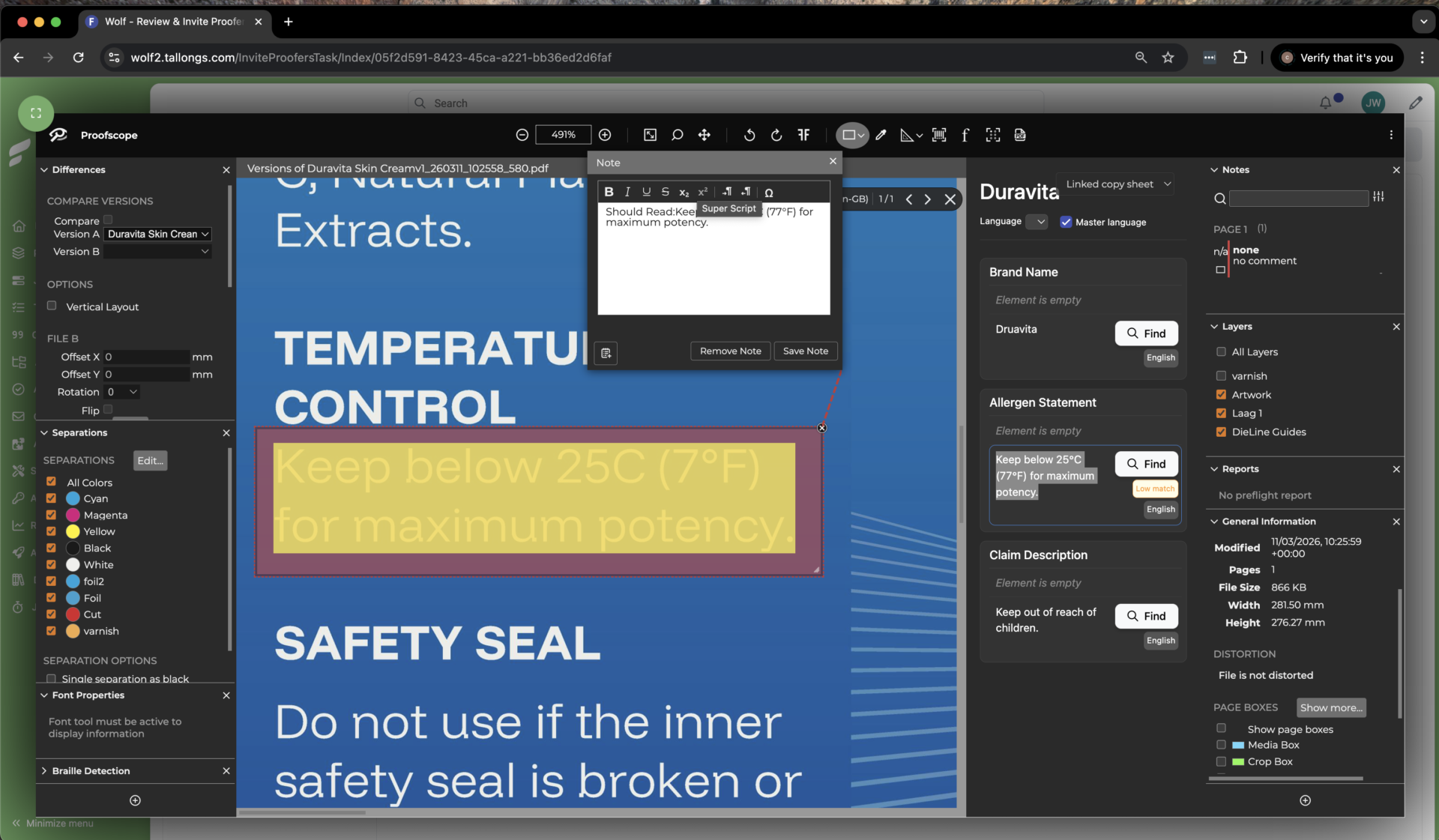
The pharmaceutical industry continues to face increasing pressure to ensure accuracy, compliance, and speed, especially when it comes to packaging and labeling. What was once considered a downstream activity is now recognized as a critical part of product integrity and patient safety. This shift is expected to be a central theme at the 15th Pharma Packaging and Labeling Innovation Forum 2026, where industry leaders will gather to explore the future of packaging, labeling, and supply chain innovation.
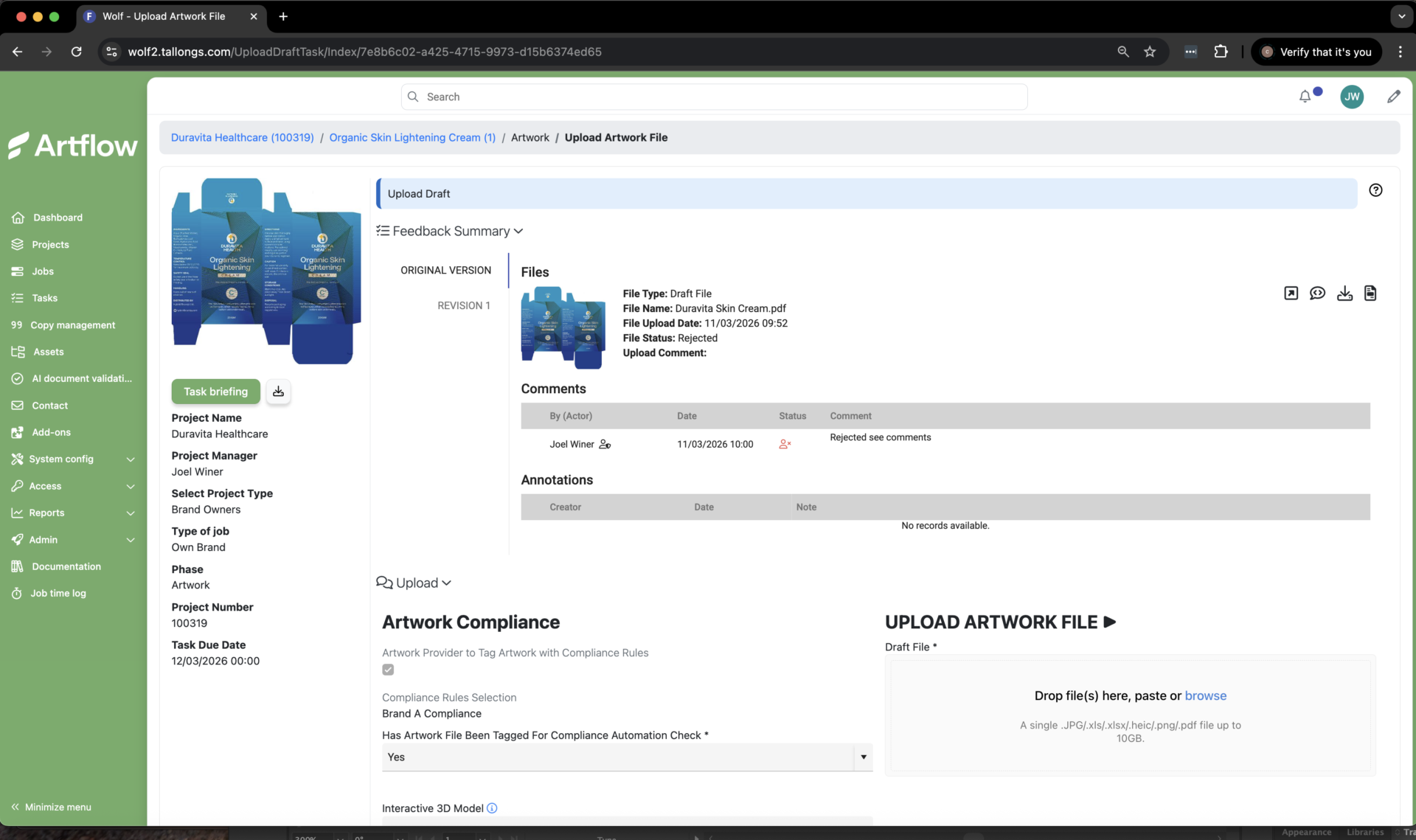
Packaging artwork uploaded into Artflow’s SaaS platform is automatically checked against integrated compliance rules, helping identify issues early in the workflow.
A key trend shaping the industry is the movement of quality upstream. Rather than relying solely on final-stage checks, organizations are increasingly investing in systems that ensure accuracy from the very beginning. This includes stronger control of labeling content and data, greater use of automation in artwork workflows, and earlier validation steps that reduce downstream risk. As regulatory requirements continue to tighten, even small errors in text, layout, or data can lead to significant delays or costly recalls, making early intervention essential.
AI is expected to play a major role in these discussions. Its ability to identify inconsistencies in labeling content, validate critical data such as dosage and compliance text, and compare versions of artwork is helping teams improve both speed and reliability. More importantly, the conversation is shifting from detection toward prevention, with AI supporting processes that reduce the likelihood of errors before they occur.
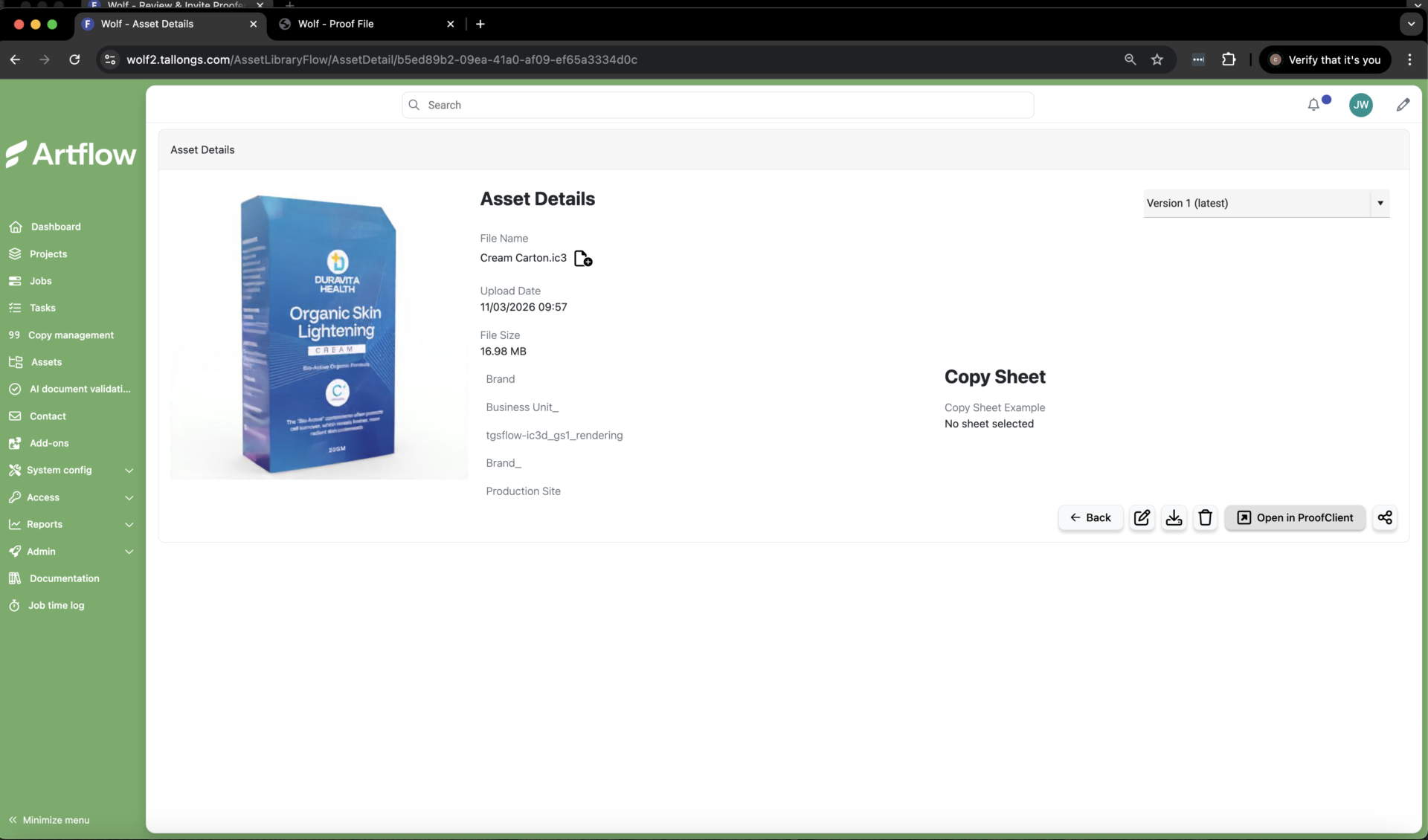
Real-time 3D visualizations powered by iC3D are generated in minutes, enabling fast and accurate proofing before production.
Another important topic is the gap between digital artwork and real-world packaging. Even when files are technically correct, issues can emerge once designs are applied to physical formats, particularly with complex shapes, materials, or finishes. Visualization and simulation technologies are increasingly being used to bridge this gap, enabling teams to preview how designs behave on actual packaging, identify potential distortions or visibility challenges, and reduce reliance on physical prototypes.
The industry is also moving toward more connected packaging ecosystems. Workflows are becoming more integrated, linking structured content and data with artwork creation, approval processes, and production readiness. This level of connectivity supports stronger collaboration across regulatory, design, and production teams while improving traceability and control.
Hybrid Brandz will be part of these conversations at USA Pharma BrainLinx 2026, sharing perspectives on how a more connected and preventive approach can be applied in practice. By bringing together AI-supported copy management and data validation, automated artwork workflows through Artflow, and real-time 3D visualization with iC3D, the focus is on enabling earlier intervention and more consistent outcomes across the packaging lifecycle.
Learn more about the event: https://usa-pharma.brainlinx.com/
Heath Luetkens, Sales Director, Hybrid Brandz Americas, speaks from 12:10-12:30 on Day, March 25th 2026, on AI-powered detects detection in pharmaceutical packaging.